
 OLEUM
ROSMARINI. U. S., Br. OIL, OF ROSEMARY
OLEUM
ROSMARINI. U. S., Br. OIL, OF ROSEMARY
Compiled and edited by Ivor Hughes
OLEUM ROSMARINI. U. S., Br. OIL, OF ROSEMARY Ol. Rosmar. [Rosemary Oil]
"The volatile oil distilled from the fresh flowering tops of Rosmarinus officinalis Linne (Pan. Labiatae). Oil of Rosemary yields not less than 2.5 per cent, of esters calculated as bornyl acetate [C10H17.C2H3O2], and not less than 10 per cent, of total borneol [C10H17OH] free and as esters." U. S. " Oil of Rosemary is the oil distilled from the flowering tops of Rosmarinus officinalis, Linn." Br.
Oleum Rorismarini, Oleum Anthos; Huile volatile de Romarin, Fr. Cod.; Essence de Romarin, Fr.,Oleum Rosmarini, P. G.; Rosmarinol, G.; Essenza di rosmarino, It.; Esencia de romero, Sp.
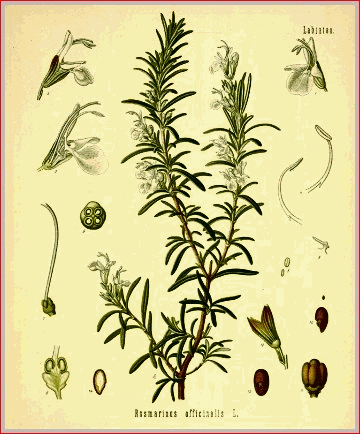
For description of the plant from which this oil is obtained see under Rosemary, Part II. The fresh leaves of rosemary yield, according to Baumĕ, 0.26 per cent, of volatile oil, but the percentage is rated much higher by others. Schimmel & Co. obtained by distillation from dry French rosemary flowers 1.4 per cent, of oil. This oil is colorless, with an odor similar to that of the plant, though less agreeable. Its sp. gr. is said to be 0.911, but reduced to 0.8886 by rectification (0.894 to 0.912, U. S.). Buignet gives the sp. gr. of the rectified oil at 0.896, and states that it is moderately dextrogyrate. Its reaction is neutral. It is soluble in all proportions in alcohol of 0.830, but requires for solution at 17.7� C. forty parts of alcohol of 0.887. (Berzelius.)
Description and Physical1 Properties.
" A colorless or pale yellow liquid, having the characteristic odor of
rosemary, and a camphoraceous taste.
" The Oil is soluble in 10 volumes of 80 per cent, alcohol, by volume. Specific gravity: 0.894 to 0.912 at 25� C. Optical rotation: varies from 0� to +15� in a 100 mm. tube at 25� C. Refractive index: 1.4660 to 1.4720 at 20� C. A solution of the recently distilled Oil in 80 per cent, alcohol (1 in 10) is neutral or only slightly acid to moistened litmus paper." U. S.
" Colorless or pale yellow. Odor that of rosemary; taste warm, camphoraceous. Specific gravity 0.895 to 0.920; optical rotation -2� to +15�; refractive index at 25� C. 1.463 to 1.473. Soluble in 1 part of alcohol (90 per cent.), and in 5 to 10 parts of a mixture of equal volumes of alcohol (90 per cent.) and alcohol (70 per cent.), Contains not less than 10 per cent, of total alcohols, calculated as borneol, C10H18O, and not less than 1.8 per cent, of esters, calculated as bornyl acetate, C10H17C2H3O2." Br.
Kept in bottles imperfectly stoppered, it deposits a stearopten, sometimes amounting, according to Proust, to one-tenth of the oil. Both Gladstone and Fl
űckiger find that most of the oil consists of a hydrocarbon, which Schimmel & Co. (Schim. Rep., Oct., 1889) identified as pinene, but there is a smaller fraction boiling at from 200� to. 210� C., which at low temperatures deposits a camphor, identified as borneol. Weber examined oil of rosemary in Wallach's laboratory (Ann. Ch. Ph., ccxxxviii, 89), and found that the fraction boiling between 176� and 182� C. contains cineol. The oil is therefore recognized at present as containing d-pinene, camphene, cineol, borneol (10-15 per cent.), and camphor. It is said to be sometimes adulterated with oil of turpentine, which may be detected by mixing the suspected liquid with an equal volume of pure alcohol, the oil of rosemary is dissolved, and that of turpentine left.Assay for Esters." Proceed as directed for esters under Oleum Menthte Piperitae, using 10 cc. of Oil of Rosemary. Multiply the number of cc. of half-normal alcoholic potassium hydroxide consumed in the saponification of the Oil by 9.811, and divide the product by the weight of the Oil taken. The result shows the per cent, of esters, calculated as bornyl acetate.
" Assay for Total Borneol.�Proceed as directed for total menthol under Oleum Mentha Piperitae, using 10 cc. of Oil of Rosemary. Use the following formula in calculating the per cent, of total borneol.
Per cent, of total borneol =
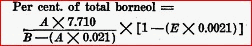
" 'A' is the result obtained by subtracting the number of cc. of half-normal sulphuric acid required in the titration from the number of cc. of half-normal alcoholic potassium hydroxide originally taken, 'B' is the weight of the acetylized oil taken, and 'E' is the per cent, of esters calculated as bornyl acetate." Preserve in well-stoppered, amber-colored bottles, in a. cool place, protected from light." U.S.
Oil of rosemary is used, chiefly in combination with other drugs, as a carminative. It is also employed as an ingredient in rubefacient liniments;. For a case of fatal poisoning, see A. J. P., xxiii, 286.
Dose
, three to six minims (0.2-0.4 cc.).Off. Prep. � Spiritus Rosmarini, Br.; Tinctura Lavandulse Composita, U. S., Br.; Spiritus Odoratus, N. F.
USD 1926 Part 11
.Rosemary
. Rosmarinus. Folia Rosmarini, s. Roris Marini. Folia, Anthos. Feuilles de Romarin, Fr. Cod. Rosmarin, Rosmariribl�tter, G. Rosmarino, It. Romero, Sp." The leaves of Rosmarinus officinalis (Fam. Labiatae)." U. S., 1880. Rosemary is a small evergreen shrub, with an erect stem, divided into many long, slender, ash-colored branches. The leaves are numerous, sessile, opposite, about 2.5 cm. long, rigid, linear, entire, obtuse at the summit, folded backward at the edges, of a firm consistence, smooth and green on the upper surface, whitish, woolly, and glandular beneath. The flowers occur in pale blue verticillasters. The plant is native to the countries which border on the Mediterranean, and is cultivated in the gardens of Europe and the United States.
The leaves, which have already been described, have a strong balsamic odor, which is possessed, but in a less degree, by all parts of the plant. Their taste is bitter and camphoraceous. These properties are imparted partially to water, completely to alcohol, and depend on the volatile oil. (See Oleum Rosmarini, Part I.) The present commercial supplies of this drug are from France. The tops lose a portion of their sensible properties by drying, and become inodorous by age. Rosemary is gently stimulant, and has been considered emmenagogue. In the practice of this country it is scarcely used; but in Europe, especially on the continent, it enters into the composition of several syrups, tinctures, etc., to which it imparts its agreeable odor and excitant property. It is sometimes added to sternutatory powders, and is used externally in connection with other aromatics in the form of fomentation. In some countries it is employed as a condiment; and its flowers, which are much sought after by the bees, impart their peculiar flavor to the honey of the districts in which the plant abounds.
Further information may be had by entering the term Rosemary into the site search box at the top right hand of the page, or else peruse the site library. Or you could try asking your questions in the site forum.
![]()