
A
TEXTBOOK OF PHARMACOGNOSY
By T.C. Denston. B.Pharm (London) F.R.I.C., F.P.S.
Compiled and Edited by Ivor Hughes
In
Two Parts.
LATICES, DRIED JUICES, AND EXTRACTS

OPIUM
Synonym: Raw
Opium.
Fam.: Papaveracece.
Official Source
Geographical
Source
Turkish Opium
Turkey, to some extent in European Turkey principally in Asiatic
Turkey (in Anatolia, a large district south of the Black Sea); exported
from Smyrna (Izmir) and Istanbul (Constantinople).
Macedonian (Salonika) Opium
Yugo-Slavia, chiefly in the South ; exported from Salonica.Also produced
in Bulgaria (in the area known as
Eastern Rumelia bordering the Black Sea) and in
Greece (in the north-east, on the plains of Thrace
contiguous with European Turkey).
Persian
Around
Ispahan, Shiraz, and Yend.
Indian
Restricted
to the central portion of the valley of the Ganges chiefly around Benares,
the opium being prepared for home use and export at a factory at Ghazipur,
44 miles north-east of Benares.
China (not exported).
Plant Habit
The opium poppy is an herbaceous annual attaining a height of about 1
metre, bearing 5 to 8 capsules. Several varieties occur, of which P.
somniferum, var. glabrum, is the one grown principally in the
Near East; its capsule is subglobular with 10 or 12 stigmata (Fig. 120),
and the petals are purple. Laticiferous vessels, though distributed
throughout the plant, are most numerous in the wall of the capsule, this
alone being used as the source of opium.
Cultivation
The plant is grown from seed, sown in porous, well-manured soil in a
sunny position. The seedlings are planted out and commence to flower in
May or June.
TURKISH OPIUM
Collection and
Preparation
1. Time of Incision. The highest yield of latex is obtained by
incision of the capsule about 14 days after the petals have dropped, the
best time for incision being indicated by the presence on the surface of a
light-green, wax-like bloom, easily removed by rubbing with the fingers.
At this stage the capsules are about 5 cm. in diameter, and are soft,
yielding to pressure by hand. The incisions are made in the afternoon and
evening, being the coolest parts of the day, in weather which consists of
clear hot days and warm dewless nights so that the exuded latex is not
washed away.
2. Method of Incision (Figs. 121 and 122). With the left hand holding the capsule, a knife is drawn lightly across the surface to make an incision extending either about two-thirds round in a transverse direction, or completely round in a spiral. These incisions must be carefully made, and must be quite shallow (usually about 1 mm. deep); if the cuts are too deep the inner wall of the capsule will be pierced, and the latex will flow inwards.
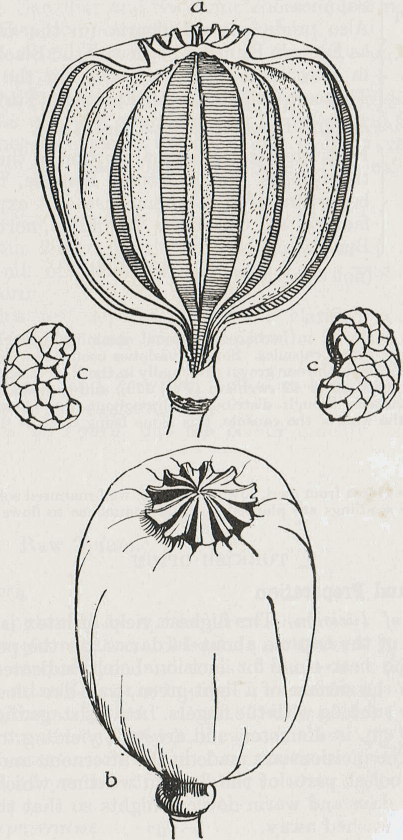
Fig. 120
(a) Poppy capsule cut
vertically. (b) Entire Poppy Capsule (c) Poppy Seeds.
UNRIPE CAPSULE OF PAPAVER SOMNIFERUM, INCISED, SHOWING EXUDATION OF LATEX;
3.
Collection of Latex. The exuded latex, which is white, turns brown
and coagulates to form a pasty mass around the incisions, and next morning
it is removed by scraping. The collector transfers the soft material to a
poppy leaf cupped in the hand, and when this is full another leaf is
placed above, and the whole is then set aside in the shade to thicken and
harden. In former years these cakes of opium were allowed to become
sufficiently firm and then placed in bags along with dock fruits to
prevent agglomeration of the cakes. The sealed bags were then enclosed in
baskets and transported by mules, chiefly to Smyrna. Here the bags were
opened, the cakes inspected, graded, sorted, and re-packed in
hermetically-sealed boxes lined with tin, called opium chests. The
product, known as "Druggists' Opium," was the variety common in
English commerce. A similar but softer variety, termed "Soft Shipping
Opium," was imported into this country for transhipment or the
manufacture of alkaloids.
 Following
the imposition of a monopoly in Opium products by the Turkish Government,
a new form of opium ("manipulated opium") has been produced
since 1935 and the old varieties ("natural opium") are no longer
imported. The inspissated latex is sent to a factory, bulked, passed
through a mill to render the mass uniform and then pressed into moulds to
give regular cakes, weighing 2 kilogrm. The cakes are coated with
coarsely-powdered poppy leaf and then packed, forty in a tin-lined case.
The product is known as Turkish Government Monopoly Opium.
Following
the imposition of a monopoly in Opium products by the Turkish Government,
a new form of opium ("manipulated opium") has been produced
since 1935 and the old varieties ("natural opium") are no longer
imported. The inspissated latex is sent to a factory, bulked, passed
through a mill to render the mass uniform and then pressed into moulds to
give regular cakes, weighing 2 kilogrm. The cakes are coated with
coarsely-powdered poppy leaf and then packed, forty in a tin-lined case.
The product is known as Turkish Government Monopoly Opium.
The collection of Macedonian Opium is similar to that of Turkish Druggists' Opium; the partly-dry juice is usually transferred from the capsules to a tin can and later made into balls of 0.5 to 1.0 kilogram., wrapped in poppy leaves, and stored until dry.
INDIAN OPIUM
1.
Time of Incision.
The time
of incision (April to June) is carefully selected as for Turkish Opium.
2. Method
of Incision.
In India,
several vertical incisions extending almost from base to apex of the
capsule are made with a sharp four-pronged instrument called a "
nushtur." Early next morning the coagulated latex is scraped off with
a small trowel-like instrument called a " seetooar " and
transferred to an earthenware pot carried by the gatherer. These are
emptied at the factory into a tilted vessel which allows a deleterious
dark fluid called " pusse-wah" to separate and drain away. The
material is then matured by exposing to the air and after reaching a
" standard " consistence, it is made into cakes or balls which
are encased in poppy leaves rendered adherent by an adhesive ("
Provision" Opium imported for the manufacture of Alkaloids), or into
cakes or square blocks wrapped in oiled paper ("Medical" Opium
for Indian Medical Department supplies or for export).
The production of Opium in India for export to China was reduced annually from 1907 by agreement with the Chinese Government, and ceased entirely in 1913. The exports from India are chiefly to this country for use in the manufacture of morphine and codeine. Indian Opium is not suitable for the preparation of official extracts, particularly Dry Extract of Opium, on account of its low morphine content, low moisture content and high water-soluble extractive
PERSIAN OPIUM
Collection resembles that described for Turkey Opium. The latex is then usually mixed with gummy matter (probably sarcocolia, a gummy exudation from species of Pendea or inferior qualities of tragacanth) and sometimes with grape juice (indicated by the high proportion of glucose frequently present to which the vitreous fraction of some specimens is attributed). This admixture is then beaten to form a homogeneous paste, and afterwards made into brick-shaped pieces which are separately wrapped in red paper.
Persian Opium is not regularly brought into this country, but quantities are exported to the Far East. Some Persian Opium is prepared in a form similar to the old Turkish '' Druggists' " Opium.
Constituents
All varieties of opium contain a large number of alkaloids, but
evaluation is normally based on the principal alkaloid, namely 'Morphine'.
12-13 per
cent in Turkish Government Monopoly Opium.
12-17 per cent in Macedonian Opium.
9-13 per cent in Persian Opium.
7-12 per cent, generally about 9 per cent, in Indian Opium.
The above figures refer to anhydrous morphine in opium as imported (raw opium). The proportion of moisture present varies and was formerly greatest in Turkish Soft Shipping (about 30 per cent) which contained 15 to 18 per cent morphine on drying. Turkish Druggists' Opium contained about 20 per cent moisture and yielded 12 to 16 per cent morphine on drying.
The Pharmacopoeia requires opium to contain not less than 9-5 per cent anhydrous morphine and this requirement is met by most consignments of the above varieties except Indian Opium, which frequently fails to reach this figure.
It has been demonstrated on a number of occasions that a hot climate is not required for the production of opium of high morphine content. Experimental cultivation in Denmark yielded opium containing 23.8 per cent morphine. Commercial cultivation, however, is not likely to develop in western European countries due to the heavier labour costs.
There
is stated to be some decline in morphine content on long storage of raw
opium but, according to Henry, powdered opium does not deteriorate in this
way when stored in well-closed containers. The alkaloids next in
importance are
Codeine.
1-1-5 per cent in good-quality Turkish Monopoly Opium.
2-5 to 3 per cent in good quality Persian Opium. 3 per cent in Indian
Opium.
Narcotine.
2 to 8 per cent, most in Indian and least in Turkish Opium, partly
free and partly as meconate and sulphate.
Papaverine.
About 0-8 per cent; after extraction of morphine and codeine from
opium, the residues are worked to extract this alkaloid. Papaverine is
also prepared synthetically on a commercial scale.
The large scale on which morphine is extracted has provided
considerable amounts of residues for the investigation of the minor
alkaloids, of which more than 20 are known. These include Thebaine 0-4 per
cent, Narceine 0-2 per cent, and the following, each of known chemical
constitution but each present to the extent of less than 0-1 per cent�
Xanthaline
(an oxidation product of papaverine), laudanine, laudanidine (also known
as tritopine), laudanoaino, oodamine, narootoline, oxynarcotine,
hydrocotamino, protopine, cryptopine, noopine (isomeric with codeine) and
porphyroxine.
Non-alkaloidal constituents of opium include;
Meconic Acid: a dibasic acid peculiar to opium and with which the morphine is combined as a water-soluble salt; the proportion ranges from 3-5 per cent, being greater in opiums of high morphine content.
Lactic acid, sulphates, meconin, gummy substances, fat, inorganic salts and caoutchouc-like material are also present.
Tannin, starch, fat and calcium oxalate are absent; they are present in a number of potential adulterants and suspected samples should therefore be tested for these substances.
Substitutes
and Adulterants.
The substances occasionally used are fig pulp, apricot pulp, and
extract of liquorice; artificial weighting is also practised by the
addition of clay, and pieces of metal, commonly lead shot. Powdered poppy
capsules have been reported as an adulterant and may be identified by the
microscopical characters of the innor epidermis of the pericarp.
Smoking opium usually contains about 5 per cent of morphine and is prepared by partially extracting opium with water, concentrating and roasting the extract.
The
evaluation of opium on its morphine content, and the care taken in
sampling for this purpose, render most forms of adulteration almost
useless; artificial weighting naturally causes serious loss, but the
practice is now rare.
OPIUM PULVERATUM. Powdered Opium (Pulvis Opii).
This
is Opium which has been dried, powdered, and adjusted to contain 10 per
cent (limits 9-5-10-5) of anhydrous morphine by the addition of powdered
lactose suitably coloured with burnt sugar, or powdered cocoa husk.
Morphine from
Poppy Straw
All
morphine produced in this country (Britain) is obtained by extraction from
opium, but in a number of European countries work has proceeded since
about 1930 on entirely different methods, based on direct extraction from
the poppy plant. The first process was devised in Hungary and patent
specifications have been published in Switzerland, France, Belgium,
Poland, England, and elsewhere. A typical process, known as the "Dry
Poppy Straw Method," uses the dried poppy straw and chaff remaining
after the ripe plants have been threshed to obtain the seed. This
"straw," which can be stored and processed throughout the year,
contains about 0-08 per cent morphine and 0-008 per cent codeine. Details
of the extraction methods vary considerably in different patented
processes.
The Opium Alkaloids and Derivatives
Morphine has been mentioned as the principal alkaloid of opium, but codeine and narcotine are also important. The mixed alkaloids are also used as Papaveretum B.P.C., containing all the opium alkaloids, in soluble form as hydrochlorides, in the proportion normally found in the drug.
Morphine. The formula C17H17NO(OH)2 for this alkaloid shows that it contains two hydroxyl groups, one of which is phenolic. For this reason two types of morphine derivative are possible, esters and ethers.
Esters of Morphine. The esters are prepared by treating morphine with acids or acid anhydrides under conditions nuitublo for esterification, and are therefore called acyl doriva.tivoH,
Like
other esters these derivatives are readily hydrolysed, whereby morphine is
re-formed�this change occurs in the body. Hence, because morphine is
readily recoverable from the esters, and because, like morphine, they are
habit-forming, all these derivatives are regulated by the Dangerous Drugs
Acts.
The more important esters are;
Diamorphine (Syn. Diacetyl-morphine, Acetomorphine, Heroin) is prepared by treating morphine with acetic anhydride, whereby both hydroxyl groups are replaced by the acetyl group (CH3CO-), as indicated by the chemical name, diacetyl-morphine.
Benzoyl-morphine. This is similarly prepared using benzoyl chloride or benzoic anhydride. This acyl derivative must be carefully distinguished from benzylmorphine (see below).
Ethers
of Morphine.
The ethers are formed by the condensation of one molecule each of morphine
and an alcohol, with elimination of a molecule of water, analogous to the
formation of the simple ethers, like ordinary (i.e. di-ethyl) ether. The
ethers differ from the esters in not undergoing hydrolysis (compare the
simple ethers in this respect). Hence morphine is not readily obtained
from them, nor is it produced therefrom in the body; consequently most are
not habit-forming. The ethers other than Codeine and Ethylmorphine are,
however, now included in the Dangerous Drugs Acts.
Ethers may be prepared from aliphatic alcohols (to form alkyl derivatives), or aromatic alcohols (to form aryl derivatives), of which the following are the more important�
Codeine. This is methyl morphine, and has the formula C17H17NO(OH)(OCH3). In addition to natural codeine extracted from opium, synthetic codeine is prepared by the tnethylation of morphine.
Ethylmorphine (Syn. Dionine). As indicated by the name, this is the ethyl ether, its formula being C17H17NO(OH)OC2HS.
Benzylmorphine (Syn. Peronine). Similarly this is the benzyl ether, and is the only commonly used aryl derivative.
In addition to the esters and ethers, there is another important derivative of morphine, namely;
Apomorphine. This is prepared by heating morphine with hydrochloric acid in sealed tubes, whereupon part of the morphine loses two molecules of hydrogen and one of oxygen, to form apomorphine, which is separated and purified. The narcotic effect of this derivative is modified by its powerful emetic properties, for which it is exclusively used.
![]()
